Health Tech Lunch - Raphaëlle Luisier - “Machine learning to transform neuronal pathological science”

The Axe Santé is organising a Health Tech Lunch on Friday, June 24th from 12:00 to 12:45 at Swiss Digital Center in Sierre (room Capella).
Raphaëlle Luisier (Genomics and Health Informatics Group, Idiap Research Institute and Swiss Institute of Bioinformatics) is a computational biologist initially trained as a bioengineer. Her research group is developing statistical and machine-learning methods to integrate genomic, clinical, and imaging data to accelerate the diagnosis and improve the understanding and treatment of complex diseases such as neurodegenerative disorders. The group has expertise in genomics, bioinformatics, RNA biology, neuroscience, data science, and data visualisation.
Machine learning to transform neuronal pathological science
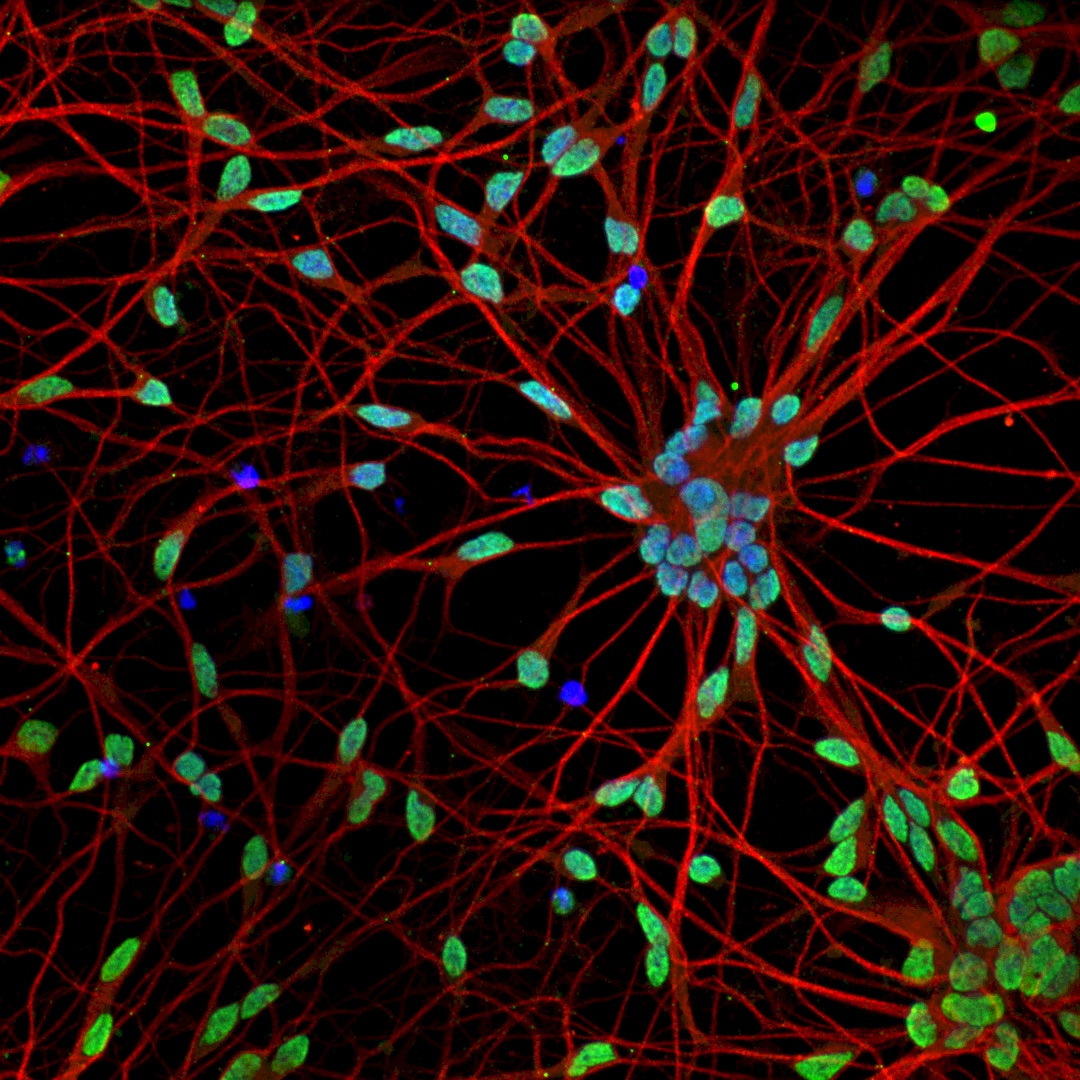
Amyotrophic lateral sclerosis (ALS), is a rapidly progressive and incurable neurodegenerative disease, characterized by the progressive death of motor neurons (MNs) for which the early cellular and molecular events remain poorly understood. We previously reported novel ALS disease phenotypes in tissue sections from mouse transgenic models, which we validated in tissue sections from sporadic ALS post-mortem tissue. These studies demonstrate that histopathological analysis of tissue sections is an invaluable resource in neurodegeneration research. Here I will present our recent developments in computational methods showcasing the striking potential of using state-of-the-art machine learning methods including deep-learning to promote innovative research directions, diagnostics and therapies. In particular I will show how combining automated image processing with machine learning methods enabled the automatic identification of sick cells with unprecedented accuracy and at single cell resolution in histopathological tissue sections, substantially improving the speed and reliability of identifying phenotypically diverse MN populations. I will next show how by adapting a pre-trained deep-learning image classifier to a rich multichannel fluorescence microscopy high content imaging data-set, we enabled the automatic and rapid generation of testable biological hypotheses and the identification of novel neurite-associated morphological perturbations in ALS disease. Our techniques could potentially be employed in a wide range of medical images across multiple disciplines, and thus might prove transformational in terms of the novel phenotypic information that can be gleaned from image analysis of in vitro neurodegeneration models.
References
1. Tyzack, G. E. et al. Aberrant cytoplasmic intron retention is a blueprint for RNA binding protein mislocalization in VCP-related amyotrophic lateral sclerosis. Brain vol. 144 1985–1993 (2021).
2. Verzat, C., Harley, J., Patani, R. & Luisier, R. Image-based deep learning reveals the responses of human motor neurons to stress and VCP-related ALS. Neuropathol. Appl. Neurobiol. (2021) doi:10.1111/nan.12770.
3. Andreassi, C. et al. Cytoplasmic cleavage of IMPA1 3’ UTR is necessary for maintaining axon integrity. Cell Rep. 34, 108778 (2021).
4. Luisier, R., Girgin, M., Lutolf, M. P. & Ranga, A. Mammary epithelial morphogenesis in 3D combinatorial microenvironments. Sci. Rep. 10, 21635 (2020).
5. Hagemann, C. et al. Automated and unbiased discrimination of ALS from control tissue at single cell resolution. Brain Pathology (2021) doi:10.1111/bpa.12937.
6. Petric-Howe, M. et al. Diminished miRNA activity is associated with aberrant cytoplasmic intron retention in ALS pathogenesis. Cold Spring Harbor Laboratory 2021.01.27.428555 (2021) doi:10.1101/2021.01.27.428555.
7. Luisier, R. et al. Intron retention and nuclear loss of SFPQ are molecular hallmarks of ALS. Nat. Commun. 9, 2010 (2018).
Programme:
Registration : https://doodle.com/meeting/participate/id/azm6mBZe
